2019-07-02 Hits(1596)
The methods available for protein purification range from simple precipitation procedures used since the nineteenth century to sophisticated chromatographic and affinity techniques that are constantly undergoing development and improvement. Several thousand proteins have been purified in active form on the basis of such characteristics as solubility, size, charge, and specific binding affinity. Usually, protein mixtures are subjected to a series of separations, each based on a different property to yield a pure protein. At each step in the purification, the preparation is assayed and the protein concentration is determined. Substantial quantities of purified proteins, of the order of many milligrams, are needed to fully elucidate their three-dimensional structures and their mechanisms of action. Thus, the overall yield is an important feature of a purification scheme. A variety of purification techniques are available.
1. Gel-Filtration Chromatography (also called molecular exclusion chromatography, size exclusion chromatography, or gel filtration chromatography) is a low resolution isolation method that employs a cool “trick." This involves the use of beads that have tiny “tunnels" in them that each have a precise size. The size is referred to as an “exclusion limit," which means that molecules above a certain molecular weight will not fit into the tunnels. Molecules with sizes larger than the exclusion limit do not enter the tunnels and pass through the column relatively quickly by making their way between the beads. Smaller molecules, which can enter the tunnels, do so, and thus, have a longer path that they take in passing through the column. Because of this, molecules larger than the exclusion limit will leave the column earlier, while those that pass through the beads will elute from the column later. This method allows separation of molecules by their size.
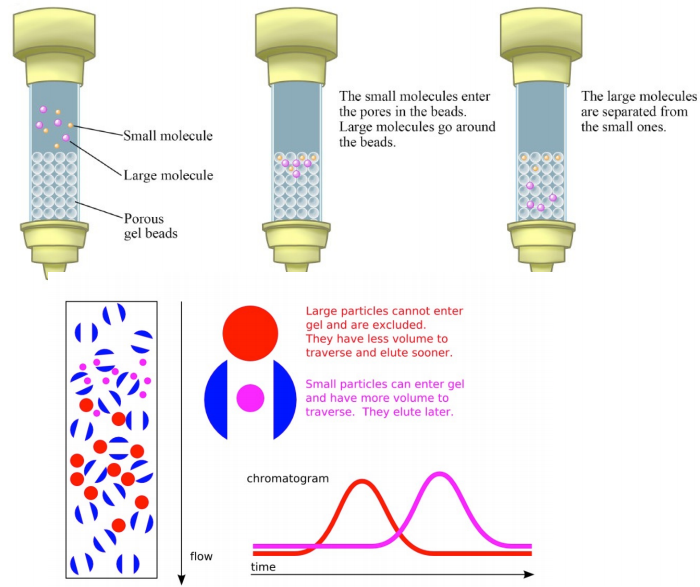
Fig.1 Gel Exclusion Chromatography
2. Ion-Exchange Chromatography Proteins can be separated on the basis of their net charge by ion-exchange chromatography. If a protein has a net positive charge at pH 7, it will usually bind to a column of beads containing carboxylate groups, whereas a negatively charged protein will not (Figure 2). A positively charged protein bound to such a column can then be eluted (released) by increasing the concentration of sodium chloride or another salt in the eluting buffer because sodium ions compete with positively charged groups on the protein for binding to the column. Proteins that have a low density of net positive charge will tend to emerge first, followed by those having a higher charge density. Positively charged proteins (cationic proteins) can be separated on negatively charged carboxymethyl-cellulose (CM-cellulose) columns. Conversely, negatively charged proteins (anionic proteins) can be separated by chromatography on positively charged diethylaminoethyl-cellulose (DEAE-cellulose) columns.
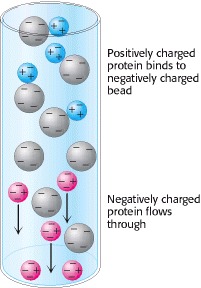
Fig.2 Ion-Exchange Chromatography.
3. Affinity Chromatography (also called affinity purification) makes use of specific binding interactions between molecules. A particular ligand is chemically immobilized or “coupled” to a solid support so that when a complex mixture is passed over the column, those molecules having specific binding affinity to the ligand become bound. After other sample components are washed away, the bound molecule is stripped from the support, resulting in its purification from the original sample.
4. High-Pressure Liquid Chromatography The resolving power of all of the column techniques can be improved substantially through the use of a technique called high-pressure liquid chromatography (HPLC), which is an enhanced version of the column techniques already discussed. The column materials themselves are much more finely divided and, as a consequence, there are more interaction sites and thus greater resolving power. Because the column is made of finer material, pressure must be applied to the column to obtain adequate flow rates. The net result is high resolution as well as rapid separation.