Service Line:+86-022-82164980
Address:FL-4, Building A5, International Enterprise Community, Tianjin, China
Email:[email protected]
Surface Plasmon Resonance (SPR) technology is an optic-based unmarked detection technology. It detects the interaction between two or more molecules in real-time by tracking changes in signals on the sensor chip. SPR signals are presented by the change of refractive index. SPR affinity assay is characterized by high throughput, high flexibility and high sensitivity, and can be used for early drug development and preclinical drug screening.
KMD Bioscience has built a comprehensive Biacore platform based on surface plasmon resonance technology, which provides fast and efficient SPR binding affinity services according to customer needs. At present, the projects we have done include various SPR binding assay between proteins and proteins, proteins and peptides, proteins and small molecules, antigens and antibodies. Our scientific research experts, for many years committed to the SPR protein protein interaction between biological molecules research, with rich technical experience, can provide customers with accurate and effective SPR screening services, to provide strong technical support for scientific research.
Surface Plasmon Resonance (SPR)
SPR is an optical phenomenon existing in nature, which refers to the occurrence of a total reflection of light on the surface of a prism and a metal film, forming an evanescent wave into the optically thinner medium, and the existence of a certain plasma wave in the medium (assumed to be precious metal). Based on the premise of conservation of energy, the resonance phenomenon may occur when the two bands meet.
With the help of traditional optical phenomena, SPR experiment uses light to generate evanescent waves in different media to generate resonance with plasma waves and thus can construct biosensing analysis technology of biomolecular interaction to detect the interaction between ligands and analytes on biosensing chips.
When the polarized light is incident on the interface of two media with different refractive indices at a critical Angle, it can cause the resonance of free electrons in the metal film, so that the reflected light is greatly weakened within a certain Angle. Among them, the incident Angle that makes the reflected light disappear completely within a certain Angle is called the SPR Angle. The specific signals of binding and interaction between biomolecules can be obtained by monitoring the dynamic changes of the SPR Angle in the process of biological reaction.
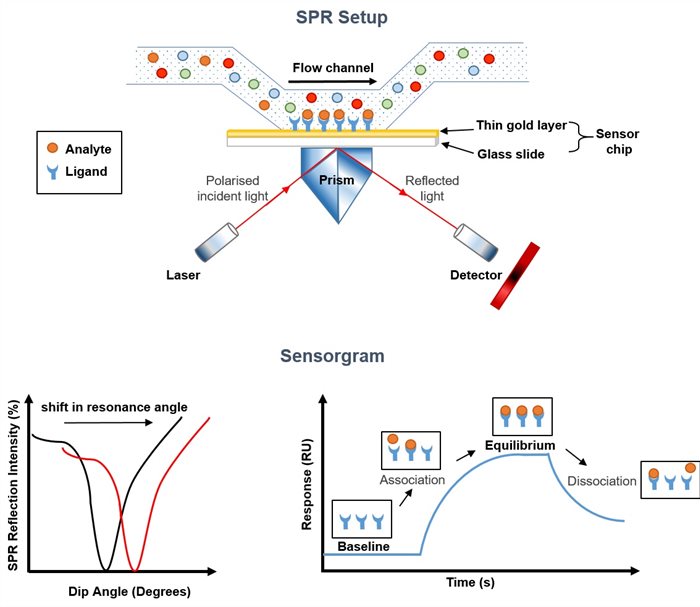
Figure 1 Schematic diagram of SPR antibody affinity provided by KMD Bioscience
Technical advantages of SPR Affinity Assay
Compared with conventional affinity detection methods, SPR binding affinity has the following advantages:
1. None of the molecules in the test need to be purified or labeled.
2. Real-time monitoring of the dynamic process of the reaction.
3. Fast results and high working efficiency.
4. It has higher sensitivity when detecting low-affinity antibodies.
SPR Affinity Assay Type
KMD Bioscience can provide SPR analysis for many types of samples, including but not limited to small molecules, proteins, nucleic acids, lipids, bacteria, viruses and whole cells. We can provide you with a variety of biomolecular information: binding specificity, concentration analysis, kinetics and affinity analysis, and thermodynamics.
SPR affinity determination is also suitable for qualitative studies to confirm the specificity of the interaction. Many successive binding events can be monitored as each leads to an increase in mass on the surface of the sensor chip and all stages of the binding process are monitored. Examples include epitope mapping, antibody isotypes and immunogenicity measurements.
Most chemical and spectroscopic methods used to quantify protein (1) measure total protein content, (2) cannot distinguish between active and inactive molecules and cannot be used in combination with unpurified samples. As SPR is a non-invasive method (no light penetrates the sample), it can measure small amounts of analyte molecules from complex substrates (e.g. food, serum or plasma and cell extracts). Direct or indirect (inhibition or competition) measurement of concentrations is possible. SPR biosensors are particularly suitable for measuring kinetic binding and dissociation rate constants through real-time measurement of binding interactions.
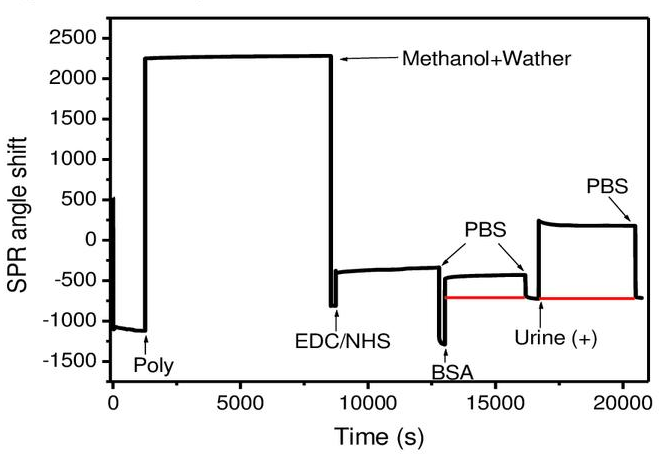
Figure 2 Bovine serum proteins adsorbed on the surface of the KMD Bioscience SPR sensor
Features of SPR Experiment
--Stable performance, for affinity determinations in specific applications, such as in the uneasy acquisition of protein, the detected affinity data is generally higher.
-- For many small molecules, the buffer requirements are more demanding and the chips are relatively expensive.
Sample Requirements of SPR Antibody Affinity
|
Customer provides |
Requirements |
|
Customer can provide antibody/protein/peptide/small molecular compound etc. |
* Buffer solution: PBS, HEPPS etc. Try not to contain glycerol, imidazole, trehalose or other salts; try to avoid reagents with amino groups such as Tris as a buffer.
* Protein: labeled > 200μg, unlabeled > 200μg, concentration > 0.5mg/ml, purity > 90%, avoiding particle or flocculent precipitation. * Antibody: > 100μg, concentration > 0.1mg/ml, purity > 90%, avoiding particle or flocculent precipitation. * Polypeptide: with biotin label > 200μg, without biotin label > 2mg, concentration > 0.1mg/ml, purity > 90%, avoiding particle or flocculent precipitation. * Small molecule compound: 1mg, concentration > 1mg/ml, purity > 90%, soluble in 100% DMSO or water. |
SPR Affinity Assay Service Highlights
-- Professional technical team and rich technical experience
-- High detection efficiency, no labeling, real-time monitoring
-- Advanced instruments and equipment
-- A wide range of applications: can be used for protein, nucleic acid, peptides, nanomaterials and other molecular interaction analysis
-- One-stop service: from affinity measurement services to molecular interaction research integrated solutions
How to Order?
If you have any questions regarding our services or products, please feel free to contact us by E-mail: [email protected] or Tel: +86-400-621-6806