Service Line:+86-022-82164980
Address:FL-4, Building A5, International Enterprise Community, Tianjin, China
Email:[email protected]
KMD Bioscience has a mature antibody production platform with advanced antibody engineering technology and extensive experience in antibody library construction. Based on these, KMD Bioscience can provide high-quality human scFv antibody library construction services and give you reliable data feedback. By using advanced phage display technology, the scFv antibody produced by KMD Bioscience ensures high affinity and stable structure。
Single Chain Variable Fragment (scFv) antibody consists of the variable regions of the heavy (VH) and light (VL) chains of immunoglobulins, connected by a short flexible linker peptide. ScFv, single chain variable fragment, smaller than full-length antibodies, offers benefits including improved tissue penetration, lower immunogenicity, flexible design for bispecific binding or fusion with other molecules for therapeutic/diagnostic use, and cost-effective production in bacterial/yeast systems.
Human scFv Antibody Libraries Construction
The phage display library is widely used in screening high-affinity antibodies, such as screening of angiogenesis marker fibronectin, tumor-specific B3/B4 antigen, and EGFR. Antibody libraries constructed by phage display technology are usually derived from RT-PCR amplification of mRNAs from peripheral blood monocytes, spleen cells, bone marrow cells, and tonsils to form cDNA fragments. These gene fragments were spliced into phagemid vectors, and then transferred into phages to form covalent structural protein expression and in vitro display. The heavy and light chains of the antibodies are expressed independently and then randomly combined to form a large antibody library with phage clone numbers up to 10^11-10^12.
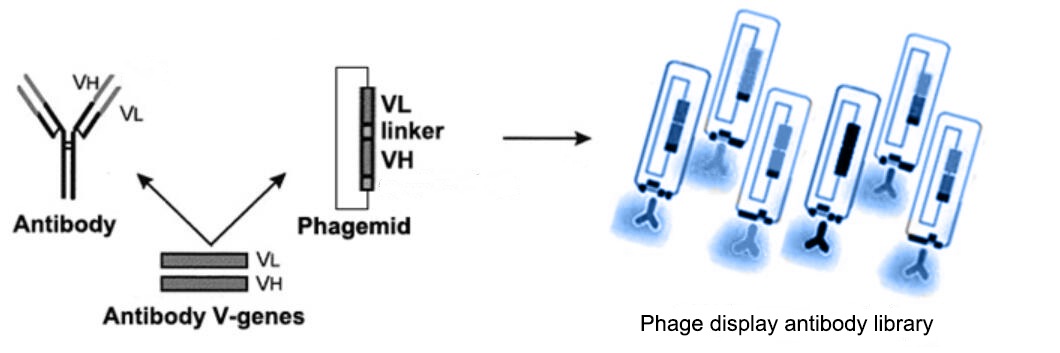
The scFv antibody library refers to the variable region (VH and VL), especially the CDR region genes of the antibody, which forms a large number of different clones by different strategies, such as amino acid mutations and frameshift mutations. And high- affinity single-chain Fv antibodies against the target antigen were screened from these clones. Compared to the Fab antibody library, scFv antibodies are more stable and less prone to form polymers due to their smaller molecular weight of about 25kDa.
Types of Phage Display System
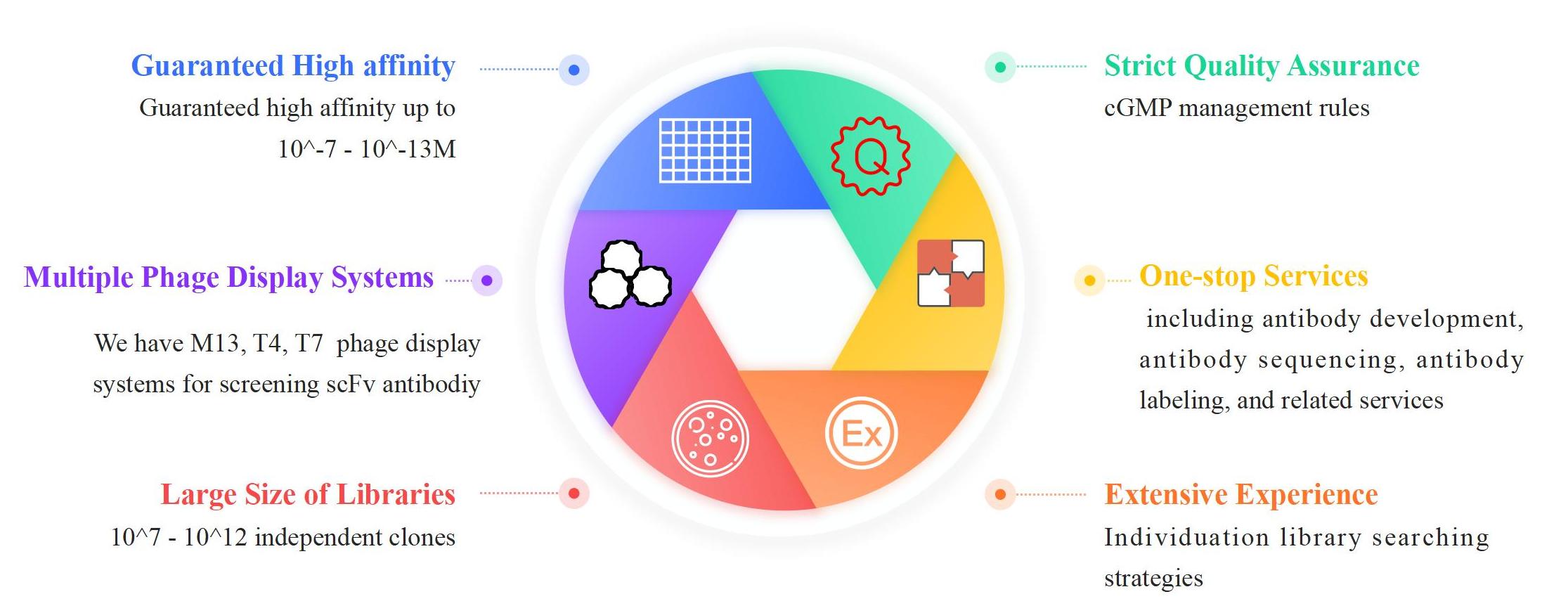
KMD Bioscience has a well-established phage display platform and has used various types of phages for different project objectives. In general, the M13 phage system is the most popular choice for phage display and is extensively used in many types of research. KMD Bioscience also provides phage display-related services via either T4 or T7 phage systems, which can provide different promising alternatives by their special merits. KMD Bioscience supports customer-tailored services. You can provide antigens, and we perform in vitro peripheral blood lymphocyte mononuclear cell sensitization to obtain higher affinity and specificity monoclonal antibodies.
Human scFv Antibody Library Phage Display Panning
Phage display panning with Single Chain Variable Fragment (scFv) antibody involves the use of bacteriophages to display scFv libraries—collections of scFvs with diverse antigen-binding specificities—on their surfaces. The screening process for single-chain antibody phages is as follows:
--1. Library Construction: An scFv library is generated by cloning variable regions of heavy and light chains of antibodies from a source (e.g., immunized animals, human B cells) into phage-display vectors. These scFvs are then expressed as fusion proteins with a phage coat protein, allowing them to be displayed on the phage surface.
--2. Phage Panning: The library of phages displaying scFv fragments is exposed to the target antigen, which can be immobilized on a solid surface (like a plate) or in solution. Only phages displaying scFvs that bind to the antigen will be retained, while non-binders are washed away.
--3. Elution and Amplification: The bound phages are then eluted, typically by changing the conditions to disrupt the antigen-antibody interaction (e.g., low pH, high salt) or by competitive elution with free antigen. The eluted phages are used to infect bacteria, where they can reproduce, generating a larger quantity of the selected phages.
--4. Enrichment and Screening: This process of binding, washing, eluting, and amplifying is repeated multiple times (usually 3-5 rounds) to enrich the library for phages that bind specifically and strongly to the target antigen. After enrichment, individual phage clones are screened for their ability to bind the antigen using techniques such as ELISA.
--5. Sequencing and Characterization: The DNA of positive clones is sequenced to identify the scFv sequences that confer specific antigen binding.
Human scFv Antibody Production Service Process
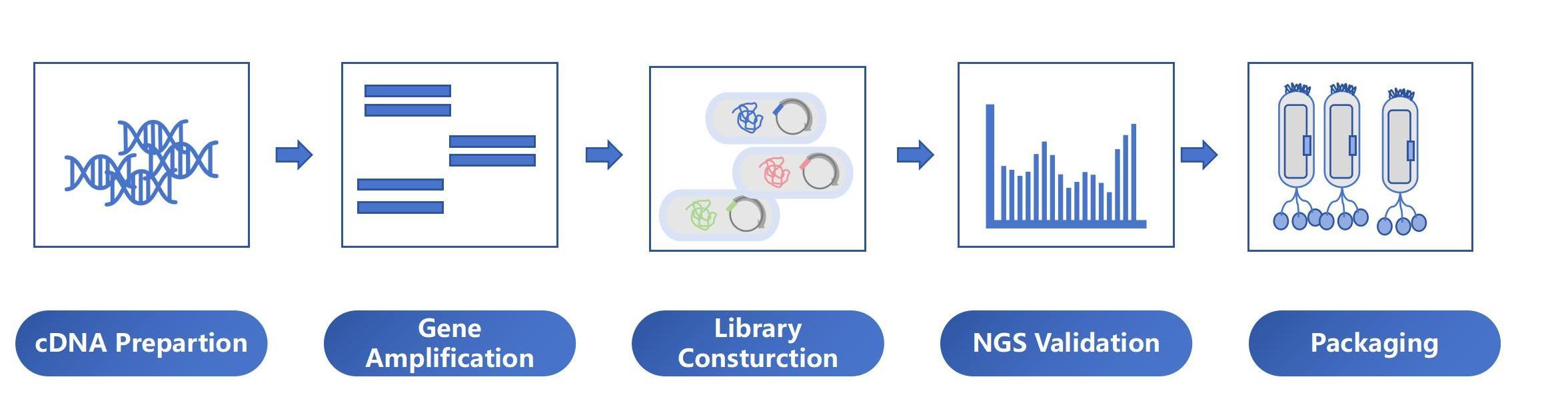
Human scFv Phage Display Service Workflow
|
Steps |
Specification |
Timeline |
Deliverables |
|
cDNA synthesis |
* Total RNA extraction. * PCR amplification with Actin-specific primers to identify the quality of cDNA. |
8-9 weeks |
* Experimental report: including detailed construction procedures and representative sequence information.
* Product delivery: including bacterial and phage antibody libraries. |
|
Phage Display Library construction |
* Primer design & synthesis * PCR amplification of variable region genes of heavy and light chains using cDNA as a template. * scFv gene splicing. * Plasmid construction & transformation: after enzyme digestion, scFv, and phagemid vector were ligated and transformed into TG1 host bacteria by an electric shock to construct antibody libraries. * Identification: 20-50 clones were randomly selected, PCR identification, sequencing, and analysis of antibody sequences. * Packaging: >1×10^13 phage particles /ml. |
Human scFv Antibody Production Service Highlights
How to Order?
If you have any questions regarding our services or products, please feel free to contact us by E-mail: [email protected] or Tel: +86-400-621-6806